|
In 2000, a pediatric heptavalent PCV (PCV7) was licensed for use in the U.S. 1 Young children and older adults are especially vulnerable, and many children who develop pneumococcal meningitis have long-term complications such as deafness or seizures. In the absence of a pediatric vaccine, pneumococcus was a significant public health concern, causing approximately 63,000 cases of invasive pneumococcal disease and 6,100 deaths in the U.S. Streptococcus pneumoniae (pneumococcus) is a major cause of invasive disease, including meningitis, pneumonia, and bacteremia. The success of the pneumococcal conjugate vaccine (PCV) in preventing suffering, disability, and death is one example that illustrates the essential role of our nation's public health systems and workforce in protecting us from vaccine-preventable diseases (VPDs). Along every step of the way, public health-at the national, state, and local levels-plays a fundamental role. But vaccination in the United States is much more complex and only made possible through a robust public-private partnership that begins with the development of the vaccine and continues long after it is used routinely. Food and Drug Administration (FDA) licenses it, health-care providers give it to their patients, and we see disease disappear. It seems simple: a pharmaceutical company develops a new vaccine, the U.S. Screening testing of asymptomatic applicants for COVID-19 as part of the immigrant medical evaluation may be required at the discretion of the panel physician.Vaccines are one of our most successful tools for protecting the public's health.Close contacts of persons with COVID-19 should also be tested and must postpone the medical examination until quarantine criteria have been met.All applicants with symptoms of COVID-19 should be tested for COVID-19 and must postpone the immigrant visa medical examination until they have met recovery criteria to end isolation.Additional information is available from the Centers for Disease Control and Prevention (CDC). Medical exams completed prior to that date will not be subject to this requirement, regardless of when the immigrant visa is issued and when the traveler enters the United States. Panel physicians will begin screening applicants for age-appropriate COVID-19 vaccinations on October 1, 2021. 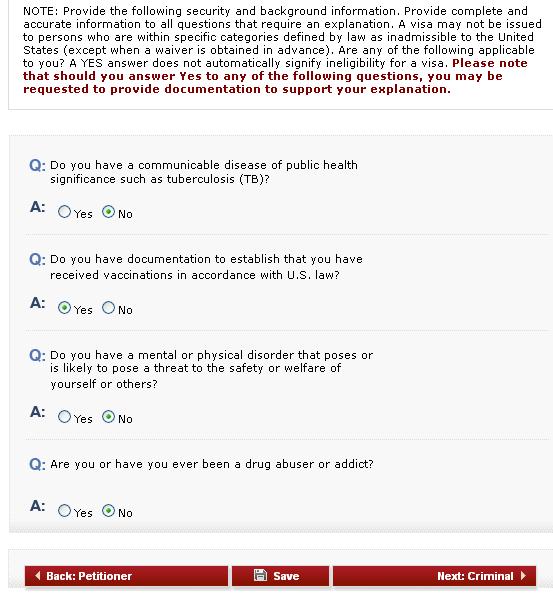
Some waivers for the COVID vaccination will still be applicable, including if the vaccine is not age appropriate. IMPORTANT NOTICE Beginning October 1, 2021, all immigrant (excluding K visas) applicants must be fully vaccinated for COVID-19 for the medical examination to be considered complete.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
